Early intervention protocol aims to reverse progression of lymphedema after surgery for breast cancer.
After breast cancer surgery, up to 40% of women may develop lymphedema, or chronic swelling of the arm. Lymphedema may be disfiguring and causes pain, heaviness, infection, and in very rare cases, severe complications such as cancer (lymphangiosarcoma). But early detection and treatment is highly effective, and the Division of Breast Surgery has established innovative initiatives to detect and prevent lymphedema in all patients treated at the Breast Center.
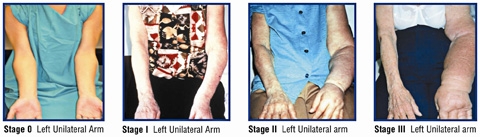
Although lymphedema is difficult to treat once it has progressed, early treatment can reverse progression of symptoms. Since 2010, the Division of Breast Surgery has maintained a unique program that includes aggressive protocols to detect and prevent lymphedema in all patients undergoing surgery for breast cancer. As part of this aggressive program, a new trial is now testing the effect of early educational intervention among patients of Chinese origin.
The Lymphedema Risk Reduction for Chinese Breast Cancer Survivors Study aims to reduce the incidence and severity of lymphedema after treatment for breast cancer through implementation of a Chinese language educational intervention. Named The Optimal You, this program emphasizes specific breathing techniques, arm exercises, proper skin care and protection, and behavioral interventions to promote lymph flow, prevent inflammation and infection, and maintain optimal body mass index. Patients will also be educated on recognizing early signs and symptoms of lymphedema so that early treatment may reverse disease progression.
Preliminary evaluation of this approach has already been demonstrated in English-speaking survivors; in the pilot trial directed by Principal Investigator Mei R. Fu, PhD, RN, ACNS-BC, FAAN, Associate Professor at New York University, 97% of women were successful in achieving improvements in lymph flow and body mass index after breast cancer treatment. Margaret Chen, MD, FACS, Assistant Professor of Clinical Surgery in the Division of Breast Surgery, is currently collaborating with Dr. Mei Fu to offer this approach to patients at NewYork-Presbyterian/Columbia.
“Based on the success of the English-language trial, it stands to reason that Chinese women will potentially benefit greatly from the opportunity to receive this intervention in their native language,” says Dr. Chen.
Patients in the trial will participate in four educational sessions, each approximately 60 minutes in duration, before surgery and at 3, 6, and 12 months after surgery. They will be asked to complete questionnaires and to undergo measurement of their upper arms to assess lymph fluid change.
In addition to The Optimal You, the Division of Breast Surgery at NewYork-Presbyterian/Columbia also offers other important lymphedema prevention protocols. All women undergoing breast cancer surgery undergo testing to measure fluid volume in their upper arms before surgery and at regular intervals after surgery. This highly sensitive, non-invasive testing (lymphedema index, or L-dex), is able to detect very early changes in lymph fluid before they are visible to the naked eye and before symptoms develop, which allows for the earliest possible intervention. The program also offers the LYMPHA procedure to reduce the risk of lymphedema for patients undergoing axillary lymph node dissection. This surgical procedure creates a bypass to restore lymphatic flow by connecting lymph vessels to a branch of the axillary vein.
NewYork-Presbyterian/Columbia is the first in the U.S. to perform LYMPHA at the time of lymph node removal in order to prevent the development of lymphedema. To date, over 30 patients have undergone this procedure with only one patient developing lymphedema.
