We sat down with vascular surgeon, Virendra Patel, MD and heart surgeon, Hiroo Takayama, MD, co-directors of the Aortic Center, to discuss the new clinical trial for endovascular total aortic arch replacement—a device called the RelayBranch System.
Using endovascular procedures to treat aortic aneurysms isn’t new, but a device or graft that replaces the entire aortic arch without opening the chest is groundbreaking. The aortic arch is the curved section of aorta that connects the ascending and descending portions. It’s also home to three key arteries: the innominate artery that sends blood and oxygen to the right arm, the left subclavian that goes to the left arm, and the left carotid artery that travels straight to the brain.
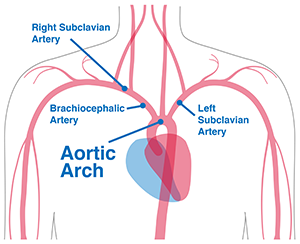
The endovascular approach is a form of minimally-invasive surgery that involves the threading of a stent graft or device through a small incision near the hip. Until now, an extensive open operation was the only way to do a total aortic arch replacement.
In this conversation, Dr. Patel and Dr. Takayama explain what makes the RelayBranch trial a milestone in the treatment of aortic aneurysms. We get into how the device works, the history of endovascular treatment, and who may benefit from joining the clinical trial.
Can you tell us a little bit about the RelayBranch trial and its significance?
Dr. Patel: Minimally invasive stuff really has revolutionized surgery, right? Incisionless surgery. So, this is sort of the cardiovascular equivalent of incisionless surgery. When we started with abdominal [minimally invasive] devices to treat aneurysms people thought it as strange and abnormal. And in fact, Juan Parodi, who's the father of endovascular surgery, was thought of as a quack, almost a radical. His original series was actually refused publication by a number of vascular journals.
Dr. Takayama: When you look back on the history, endovascular repair for aortic aneurysm only started about 30 years ago. And it started with replacing smaller parts of the aorta farthest away from the heart. The first type of this device was able to reconstruct only the left subclavian artery, the most difficult branch. So, it was only available if you had an aneurysm in that artery. Then, basically, we started from the distal [farthest away] and began marching proximal [nearer] over time.
Wow. When did the endovascular approach start to catch on?
Dr. Patel: The first study was in the 90s, and it was a series of six patients where he [Parodi] stented aneurysms and showed that it could actually be done safely. And then from there came companies that thought of this as revolutionary and as a big business. They developed devices, and it took off to the point where now 70 to 80 percent of all aneurysms in the United States are fixed endoscopically.
So, tell us about the gradual march toward total replacement. How did progress move?
Dr. Patel: As Hiroo mentioned, we started with the easier, less dangerous stuff. Then we moved to stent devices in the thoracic aorta (which is also a straight tube). Interestingly, similar to Parodi’s experience, people started doing those devices based on what the experience in the abdominal was [with other minimally invasive devices], and surgeons were making their own grafts. The first published series had 15 patients in it. And they found that 14 of the 15 patients had complete cure of their isolated thoracic aneurysms.
 Dr. Takayama: From there we were able to start reconstructing arteries one and a time, gradually, endoscopically, moving toward the main part of the arch with the development of technology. Anatomically, the device became more and more available to move north toward the heart. The arch itself has been one of the most difficult repairs because it has branches to the brain.
Dr. Takayama: From there we were able to start reconstructing arteries one and a time, gradually, endoscopically, moving toward the main part of the arch with the development of technology. Anatomically, the device became more and more available to move north toward the heart. The arch itself has been one of the most difficult repairs because it has branches to the brain.
Dr. Patel: And then came the pioneers who said, “Okay, well we've got devices from below the visceral segment.” The visceral segment is where the renal [to kidneys] and the mesenteric arteries [to the gut] are, and then the thoracic aorta is the straight tube without branches. The pioneers said, "Well, why can't we make a hole?" And so, the early folks started burning holes. Maybe started with one renal artery and they said, "Well if I can do one, why can't I do two? If I can do two, I can do three. If I can do three, why can I do all of them?" And so that's how these devices became available for the visceral segment. That's how people like myself started doing certain modified grafts for two, three patients that we thought were too high risk for open surgery.
Right now, without this trial, what happens in open surgery? Hows does open repair work?
Dr. Patel: Currently the only thing available is a median sternotomy [where the sternum is split] and circulatory arrests, which basically means “cold and dead.” We stop the heart under really low-temperature conditions so that the body is preserved by the cold and then start everything back up when you're done. It takes about 7 hours to complete.
Dr. Takayama: For the main part of the arch that involves the proximal two branches, we had to treat those aneurysms using a variety of adjunct therapies—for instance, debranching graft devices. With those head vessels, if we create a different route for blood to flow into the brachiocephalic artery and left subclavian artery from the ascending aorta [instead of the arch], we can place an endovascular graft across the aortic arch without compromising blood flow to the brain. Because that artery was rerouted with debranching grafts. But, it requires open surgery to do the debranching procedure. This device technology, the RelayBranch, eliminates that.
Dr. Patel: Right, it’s a way to do this now without any circulatory arrest, to do it minimally invasively. And I mean we still going to have to make some incisions to protect the brain, to deliver some of the stents, but it's a much smaller operation. It's a much faster recovery.
This RelayBranch trial is called a feasibility trial, what does that mean exactly?
Dr. Patel: There are feasibility and pivotal trials. The feasibility trial is the test-water trial where the FDA will say, “10 procedures in the country. That's it, and then we'll reassess.” So on the original feasibility [trial], there were 10 or 12 patients, which were all done safely. And now they're doing what's called expanded feasibility, which was going to probably double the number. There will be at least 30 patients worth of experience or data for the FDA to say, “Okay, this is safe.” And now you can do a feasibility study or a pivotal trial. And a pivotal trial would test for safety and efficacy at 30 days, 1 year, 5 years.
So this is different from a randomized control trial?
Dr. Patel: Correct. Unfortunately, with a device like this, you're not going to be able to flip a coin to truly do a randomized controlled trial, which is the gold standard to say, “Okay, this patient's going to get an open procedure and this patient's going to get the endovascular graft.” That's not going to happen. Once you give people a choice, then everyone's going to want them minimally invasive if it's safe for them.
What is the endoscopic device made of? Is it similar to the graft used in open procedures?
Dr. Patel: It’s the same fabric material [Dacron] we use for our open reconstructions except now it has a metal cage sown in to help it expand.
Dr. Takayama: When we do it open, we attach the graft to the artery and so it's actually the suture line that opens the graft. And then the blood flow pressurizes it and it stays open. But with this device, blood pressure wants to crush it. Blood pressure will crush it, so the stents actually have to pop open and then friction holds onto it throughout its course.
How much quicker is this endoscopic method, surgically?
Dr. Patel: We would be able to do this in about 1-2 hours. Probably for the first couple it's going to take us a little longer, but when Hiroo and I actually did our first practice test in a beating heart model it took us less than an hour to do the procedure.
Wow, that’s amazing. What will recovery be like?
Dr. Patel: Recovery would be two small incisions and a large puncture in the groin. The biggest risk of a device like this, because you're passing large hardware across the arch which feeds the brain, are small bits of debris on the device or air particles in the device that could cause strokes. So, that's going to be the Achilles heel. But in a patient who doesn't have strokes or stroke risk, whose operation is done within the hour, hour and a half or so that it would take us, they could be home the next day.
How many full-arch repairs do you do right now? How many open procedures per year could this affect?
Dr. Patel: We do about 20 to 30 a year.
Would all of them be candidates for the endoscopic procedure? Aside from those with stroke risk?
Dr. Patel: Yeah, potentially all of them.
Dr. Takayama: This will open the door immediately to those patients who are very high risk for open surgery. The truth is many of the patients who need heart surgery are high risk, so this is excellent news, really.
Other than the obvious benefits of the surgery itself, is this device more durable than grafts used in open operations? Are there additional benefits?
Dr. Patel: Durability may be an issue, we don’t know. We do know for other grafts that 5 to 10 years is probably a reasonable lifetime, at which point the fabric can have some wear and tear, devices may need to be realigned. So, this may open up a whole set of different problems in the future. But infrarenal grafts [below the kidney] require long-term observation.
I think that's why we have to leave these devices in the hands of experts who do both open surgery and endo and will choose what’s right for the patient. If you look at what's happened in the infrarenal space, the entire country has shifted to being minimally invasive, non-open surgeons. And so oftentimes they make the wrong choice for a young healthy patient, who probably needs a 20 or 30 years solution not a 5-year solution. I, in my own practice, am seeing more and more of these failed grafts that have to be redone.
It seems that the most significant part of this treatment is that it’s very individualized, especially in selection.
Dr. Patel: It should be individualized, but it won't be. Once these devices are fully available to the masses, they're going to be put in haphazardly, I think. Or without the appropriate clinical decision making.
Are there other risk factors besides stroke that would keep someone from being a candidate for the RelayBranch Trial?
Dr. Takayama: Yeah, we're looking at disease in the arch, disease in the vessels, so this procedure then involves the aortic arch, the ascending aorta, the descending aorta and at least two out of three of the head vessels. I think in the future there will be a three head vessel device, which would make things even more simple. Currently, we at least have to deal with the artery to the left arm with a short operation ahead of time so that all of the blood vessels get perfused.
Dr. Patel: We want to make sure that we pick the right patients. So anyone that has a disease in any of those vessels, disease in the arch, or if they have a big aneurysm that's full of debris, and we think that passing this device through the arch is going to knock something loose and potentially put them at risk of stroke—those patients probably shouldn't be done this way. Those patients should be done with open surgery, with all the things we have during open surgery, to protect the arteries by shutting off the flow to the brain. If we knocked something loose, we see it right then and there and we can clean it out.
How far away are we from this device receiving full FDA approval? How quickly will we reach this point of mass availability?
Dr: Patel: I think it's going to be kept very limited in the U.S., and we are probably 5-7 years away from a full FDA-approved arch device that's commercially available. After probably 5 years of availability of these kinds of devices in Europe and South America, this is the first trial in the United States and it's limited to 10 centers. And there's probably going to be heavy training requirements and skill set requirements, or team dynamic requirements before it's allowed to be publicly utilized.
Is that how the Aortic Center came to be one of the 10 centers included in the trial? How does that process work?
Dr. Patel: Bolton, the device company, picks centers based on volume of outcomes and the dynamics of the team. With a device like this, a lot of these patients would most likely be seen by people like Hiroo in CT surgery, but a lot of the skills required for this are a very daily part of my practice. If I had the patients, sure I could do it, but Hiroo has the patients and he also has the expertise for when things go wrong towards the heart. And I have the expertise for when things go wrong towards the other end of the graft.
Dr. Takayama: Companies say that they pick centers that have this collaborative effort. That's one of the selection criteria—expertise in both cardiac and vascular surgery and collaborated team effort. Working in a team.
Dr. Patel: We're actually deciding, collaborating, working together in the operating room, and sharing the expenses and the profits. We don't have a turf war. Patients come and they get the team, and we give them what we think is best for them in terms of the procedure, not because my division's going to make more money doing procedure B versus this procedure. And that happens. Money makes the world go around, unfortunately. And in most institutions, it's all about the battles over money.
Speaking of money, how does money factor for patients?
Dr. Patel: The way it works in the U.S. is that companies have to petition the FDA and put up the expense of the trial. All of the devices—the device itself—that are done in the trial are free. But when they sell them to hospitals in the UK or elsewhere, they're getting paid $25,000. A lot of these devices that come to the United States are really expensive. These trials would cost hundreds of millions of dollars to get FDA approval. And if the patients are poorly chosen, and there's a high stroke rate, and the devices or the trial is shut down, it’s a huge investment down the drain and we can’t use it here.
And that could happen even after it’s already being successfully used outside the U.S.?
Dr. Patel: Correct. And still be available, successful, in Europe to all Europeans and everyone outside the United States. That’s why the key is to have a huge practice with good outcomes, good decision making and as Hiroo said, we work collaboratively. What if we only saw 1 or 2 of these a year? We wouldn’t and shouldn’t be in the trial.
Does that mean that there’s already a lot of data on this device?
Dr. Patel: It's very frustrating because the Europeans have data on hundreds and hundreds of patients and experience that shows which patients should be ideally selected for, and now we're starting all over. And I think that data is what the companies used to show the FDA that there's a basis, while experimental, that the device has the potential to benefit patients. But I mean, why should we have a 5-10-year delay for devices that are already being used and benefiting patients elsewhere?
Do you think there’s any potential that the approval process could change?
Dr. Patel: There are fast track processes, certainly for things like cancer drugs for highly lethal cancers. But for devices, unless they are highly lethal, not really. I mean, the aneurysms are lethal and maybe this is the fast track version of the process. I can't imagine what it would be like for a slower process because we're already beating our heads against the door to get access to these things. And we’re finally here.
For someone who wants to learn more about the trial and thinks they could be a candidate, who should they contact?
Dr. Takayama: Reach out to us through the aortic site, or phone number: (212) 305-6380.
Dr. Patel: Yes, contact us. The Bolton website has a lot of information about endovascular devices and you can look up literature on the arch graft trials at NIH. The trial is now up and running!

