I-SPY Trial Is Changing the Standard of Treatment for Locally Advanced Breast Cancer
“The right drug. The right patient. The right time. Now.”
That’s the refrain from researchers and clinicians of the groundbreaking I-SPY trials, a neoadjuvant treatment [when medications are administered before surgical intervention] for locally advanced breast cancer. This isn’t your average trial. Instead of employing the traditional model for randomized trials, this is an 'adaptive' model. A design that has amped up efficiency for evaluating experimental agents.
That efficiency has led to a rather surprising and groundbreaking discovery for those who receive neoadjuvant therapy before surgical treatment: there is no difference in recurrence rates between those who undergo mastectomy versus those who undergo lumpectomy. Meaning, the data shows no benefit to removing additional breast tissue. A huge development, and surprise to researchers and clinicians alike.
So, what does this all mean, exactly? Will mastectomy soon be a thing of the past? We spoke with Chief of Breast Surgery, Roshni Rao, MD, about what you need to know.
Let’s start with a brief overview of the study. What is I-SPY?
I-SPY is a national consortium, and it's really an innovative trial because they use a combination of MRI and neoadjuvant treatments, which means treatments that we give before breast surgery, to really try and sort out what medications and what therapeutics are potentially most effective for specific tumor types.
Traditionally, we’ve gone through these huge, randomized trials, it takes a long time, and it costs a billion dollars. With I-SPY, we have been able to utilize MRI as a tool to predict response, look at different tumor types, and take the most promising agents out of preclinical trials—all of these agents have some sort of data behind them. Then give patients either the standard or the standard plus this medication. So, it's a really innovative way to quickly parse out which medications might not work in the neoadjuvant setting by using MRI findings.
From this collection of promising agents, how are the medications and therapeutics selected for participants? What does the MRI show?
What we do is figure out what type of cancer a patient has based on the biopsy and tests—and right now, this is only for bigger tumors. Then we profile the tumor and say, "Okay, these are a couple of new medications that we think, based on some preliminary data, should work for your tumor.” Next, you’re going to get an MRI and be given the medication, if you’re randomly selected for it. And we're going to analyze your response as well as MRI findings to see if it works.
A lot of the immunotherapies and some of the other medications for breast cancer came through this trial. It’s already had a lot of impact because it’s been open for over a decade now.
In the future, is the goal that these treatments would be applied to earlier stages of cancer, smaller tumors, too?
Yes. The concept is that you prove these therapies work in the neoadjuvant setting. And then you can, of course, add it afterward in the post-adjuvant setting [after surgery or main treatment] as well.
Here's how researchers describe the process:
A new participant’s breast cancer is classified into one of 10 molecular subtypes. Then:
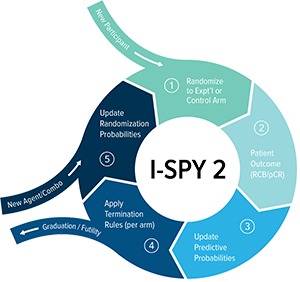
- A participant is randomly assigned to a study arm. Greater weight is given to arms that have been successful in your tumor subtype.
- The endpoint is assessed at the time of surgery. The primary endpoint is what’s called pathologic complete response, meaning the tumor has disappeared completely.
- Data based on a participant's tumor subtype, outcome, and treatment received, is updated to the predictive probabilities engine in real-time.
- If predictive probabilities for an experimental agent reach efficacy, it is declared a success. Alternatively, it may be stopped for futility. At any point, new agents may be added.
- The participant's data continuously refines the targeting of subsequent participants. Making the entire trial increasingly effective over time.
So, in the neoadjuvant process of this trial, does it potentially change the treatment trajectory entirely?
It does. And we’re doing these additional de-escalation approaches as well more recently. We're giving the least toxic regimen that we think could work and then doing a needle biopsy of that area to see if they've got what we call a pathologic complete response.
We give that chemotherapy, do a core biopsy of it, and if there are no more live active tumor cells left, that's great, and you do the surgery you had planned. But if they do that biopsy and there are still a few tumor cells left, we bring in the more aggressive chemotherapy agents. Because obviously, you've proven that those patients need the more aggressive treatments.
What are some of the differences between these chemotherapy agents, other than strength of toxicity? Are any particularly promising?
One of the things that we struggle with are big tumors that are estrogen and progesterone receptor-positive. We don't like to give those patients chemotherapy unless we really need to, but sometimes we need to shrink the tumor to get them to be able to have a lumpectomy or see what the response is. There are a few agents now available through I-SPY that are targeting that specific tumor type to see if we can find some way to shrink those nicely.
Interesting. What kind of shrink response are you seeing in those tumors so far?
It depends on the tumor type, certainly. But when I started in practice, we used to tell people that we'd get pathologic complete response 15 percent of the time [Pathologic complete response, or pCR, means that no remaining cancer cells are seen when the breast tissue is examined under a microscope]. Now being able to really target tumors appropriately, routinely, we're seeing 40 to 60 percent pathologic complete response rates. So much, much better.
Because of the way this trial works, with new and different agents being added, removed, and tested all the time, will I-SPY continue to expand for the foreseeable future?
Yes, I-SPY will continue. It’s almost an infrastructure to get clinical data for these promising agents, using MRI findings and response, etc. So, it's going to keep going and become a really rapid way to get data on what's promising. This study looks at nearly 1,500 patients within I-SPY, who underwent neoadjuvant treatments. The patients then either had lumpectomy or mastectomy, and it does an analysis on that. Traditionally, we've always told patients that if you choose a lumpectomy, you might have a slightly higher risk of recurrence, but the survival is the same.
This study shows that with this approach of neoadjuvant response, then doing a lumpectomy, and then doing radiation for the patients who need it, there's no difference in recurrence. And I think that surprised all of us!
Wow. What does that mean for patients choosing lumpectomy vs. mastectomy, exactly?
That's what's really exciting about it. We’ve known for years that just because you chose a more aggressive surgery, mastectomy doesn't improve survival. With lumpectomy, there have been studies that showed it increases local recurrence. And inherently, it makes sense, right? If you're leaving most of the breast tissue there, you would think your risk of local recurrence would be higher. So that's been what we always tell patients—that your survival won't be changed if you choose a lumpectomy, but you might have a higher risk of the cancer coming back in the breast. That chance has been enough for some to say, "No, I never want to come back. I want a mastectomy." But this study shows that there is no advantage to mastectomy; it’s equivalent.
Does that mean this neoadjuvant approach could potentially eliminate the need for mastectomy in a lot of cases?
Yes, I think for patients who respond appropriately, it could. And it changes the conversation that we have with patients. I think the options of lumpectomy or mastectomy will still be there, and it will be up to the patient. But rather than telling patients that the local recurrence is going to be higher with lumpectomy, for patients who got neoadjuvant treatments, it seems like the local recurrence is not higher.
So, after so long, the guidelines for treatment are changing? That’s pretty amazing.
Yeah, and I think patients should feel comfortable choosing a lumpectomy. That's another thing that's out there—patients feel like more surgery should be better. "If I remove the whole thing, it's got to be better." But this very solid, nationally-done trial is showing that is just not the case, and I think we really need to get that fact out there.
Are there situations, say, people who carry genes like BRCA, where mastectomy would still be advantageous?
Yes, that’s completely different. There are all types of breast cancers, but in I-SPY, we don’t specifically collect the genetic testing results as part of the study. Most of the time, when patients are diagnosed with a gene, generally, those patients do not choose lumpectomy. So, we don't have the level of information to determine if there would be a difference between mastectomy or lumpectomy in these cases.
But for those who are not gene carriers, undergo chemotherapy, and have a good response, there's no reason for them to feel like mastectomy is better.
What do you expect in the future? Does this trial change possibilities for breast cancer treatment moving forward?
Absolutely. There are some preliminary studies that look at the concept of giving these therapies, doing the biopsies, and if there's a great response, either not doing surgery and just doing radiation, or just surgery and not doing radiation.
I think that's the next step. And I-SPY is trying to sort out the question: Do we really need to do chemo, surgery, and radiation for every person? Or if someone has a good response, could we do surgery or radiation? We’re really trying to nuance out what the local treatment needs to be and de-escalate the things that maybe a patient doesn’t need.
Sounds like a pretty big deal.
It is really exciting. I think many times we're at our own cancer centers trying to do our things, but you can only get this level of data when everyone works together as a consortium. Where we can get large numbers of patients and real-world data. There's no one center that's going to cure cancer alone. It's got to be these real collaborations that push us forward.
To make an appointment or speak to a specialist about trial participation, please call (212) 305-9676 or fill out our appointment request form.
For more information on I-SPY, and clinical participants near you, click here.
Further Reading:
- Oncoplastic Surgery: An Interdisciplinary Trend Towards Breast Conservation in Breast Cancer Surgery
- Reconstructive Surgery is a Life-Changing Process of Innovation and Integrative Care
- State of the Union: Breast Care
Sign up for our mailing list for more health news and information from Columbia.

